HIV protection now becomes affordable with two landmark deals.
Sept 2025 : Today, two partnerships were announced that provide millions of people in developing nations with access to what is widely described as the most effective HIV prevention drug at an annual cost of just $40 per patient, which is just 0.1% of its original cost.
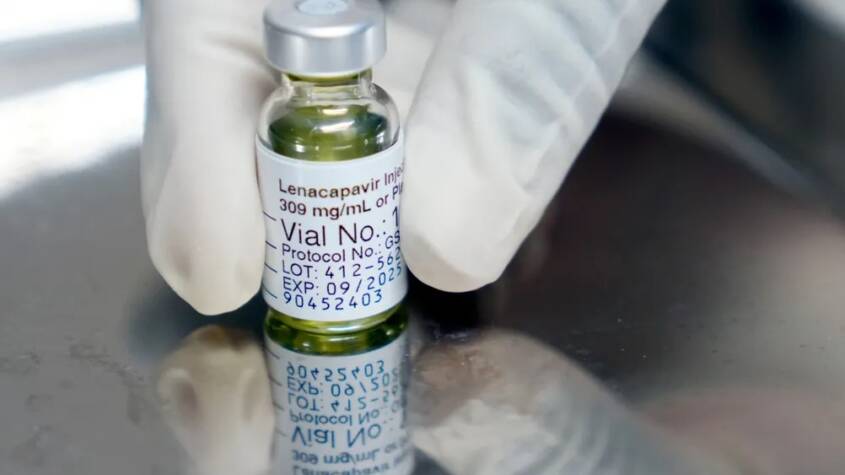
The new drug, Lenacapavir, is administered through a single injection every six months and has been shown to provide near-perfect protection against the virus. Under the terms of two agreements between global philanthropic organisations and Indian pharmaceutical companies, Lenacapavir will be made available to people in more than 100 low and middle income countries starting in 2027.
The agreement announced on Wednesday (24th. September, 2025) was reached between the Clinton Health Access Initiative in partnership with the Gates Foundation and other groups, including South African research institute, Wits RHI. “For many low- and middle-income countries, affordable access to HIV prevention is not a luxury, it is a necessity“, Professor Saiqa Mullick from Wits RHI told the Press. She added that Lenacapavir had “the potential to transform prevention, especially for young people and underserved communities who struggle with frequent clinic visits.
“The real work begins now, partnering with communities and governments to build demand, secure commitments, and prepare systems so countries are ready for rapid introduction and scale“.
Lenacapavir has delivered impressive trial results and, in July, it received official backing from the World Health Organization for HIV prevention. The injection is taken twice a year and provides six months of protection against HIV infection at a time.
Experts say long-acting injectables like Lenacapavir could help reduce new infections in populations that are most vulnerable, including adolescent girls and young women, LGBT people, sex workers, and those who use drugs. It is hoped it will replace the current form of HIV preventative drug, known as PrEP, or pre-exposure prophylaxis, which is taken orally and also costs $40 per person each year.
The pills are taken daily, which can be difficult for patients to consume, and can carry stigma in certain societies. The daily dose also makes it harder to access consistently. According to the Gates Foundation, only 18% of those who could benefit from PrEP currently have access.
Lenacapavir has already been approved by the US Food and Drug Administration and the European Commission this year. Last year June, the US drug company Gilead announced that a trial of its HIV drug Lenacapavir had a 100% success rate. The new, generic version is still pending regulatory approval but it is hoped it will be available within 18 months.
One study says that increasing access to the injection to just 4% of the population could prevent up to 20% of new HIV infections. The drug can be used to protect people from catching the virus, but also to treat those who have it, according to scientists.
The announcement comes amid a turbulent year for global health, particularly the fight against HIV and Aids, following swinging cuts by the US government’s foreign aid programme, USAID, under the Trump administration.
According to UNAIDS, more than 40 million people are currently living with HIV. Despite huge strides made to reduce global HIV rates and AIDS-related deaths since 2000, about 1.3 million people still contracted HIV last year, and more than 600,000 people died from AIDS-related illnesses.
South Africa remains the country with the highest number of HIV cases, with almost eight million people living with the virus. It will be one of the countries to access the new, cheaper drug. South Africa’s department of health told the Media that it “supports any efforts to make life saving treatment like Lenacapavir available and accessible at affordable rate to all countries, including low and middle income countries, as and when they need it“.
A large clinical trial in South Africa and Uganda has shown that a twice a year injection of a new pre-exposure prophylaxis drug gives young women total protection from HIV infection.
The trial tested whether the six-month injection of lenacapavir would provide better protection against HIV infection than two other drugs, both daily pills. All three medications are pre-exposure prophylaxis (or PrEP) drugs.
Physician-scientist Linda Gail Bekker, principal investigator for the South African part of the study, tells Press Reporters what makes this breakthrough so significant and what to expect next.
The Purpose 1 trial with 5,000 participants took place at three sites in Uganda and 25 sites in South Africa to test the efficacy of lenacapavir and two other drugs. Lenacapavir (Len LA) is a fusion capside inhibitor. It interferes with the HIV capsid, a protein shell that protects HIV’s genetic material and enzymes needed for replication. It is administered just under the skin, once every six months.
The randomised controlled trial, sponsored by the drug developers Gilead Sciences, tested several things.
Firstly, whether a six-monthly injection of lenacapavir was safe and would provide better protection against HIV infection as PrEP for women between the ages of 16 and 25 years than Truvada F/TDF, a daily PrEP pill in wide use that has been available for more than a decade.
Secondly, the trial also tested whether Descovy F/TAF, a newer daily pill, was as effective as F/TDF. The newer F/TAF has superior pharmacokinetic properties to F/TDF. Pharmacokinetic refers to the movement of a drug into, through, and out of the body. F/TAF is a smaller pill and is in use among men and transgender women in high-income countries.
The trial had three arms: Young women were randomly assigned to one of the arms in a 2:2:1 ratio (Len LA: F/TAF oral: F/TDF oral) in a double blinded fashion. This means neither the participants nor the researchers knew which treatment participants were receiving until the clinical trial was over.
In eastern and southern Africa, young women are the population who bear the brunt of new HIV infections. They also find a daily PrEP regimen challenging to maintain, for a number of social and structural reasons. During the randomised phase of the trial none of the 2,134 women who received lenacapavir contracted HIV. There was 100% efficiency.
By comparison, 16 of the 1,068 women (or 1.5%) who took Truvada (F/TDF) and 39 of 2,136 (1.8%) who received Descovy (F/TAF) contracted the HIV virus. The results at a recent independent data safety monitoring board review led to the recommendation that the trial’s “blinded” phase should be stopped and all participants should be offered with PrEP as a choice.
While PrEP is not the only prevention tool; It should be provided alongside HIV self-testing, access to condoms, screening and treatment for sexually transmitted infections and access to contraception for women of childbearing potential.
In addition, young men should be offered medical male circumcision for health reasons.
But despite these options, we haven’t quite got to the point where we have been able to stop new infections, particularly among young people.
For young people, the daily decision to take a pill or use a condom or take a pill at the time of sexual intercourse can be very challenging. HIV scientists and activists hope that young people may find that having to make this “prevention decision” only twice a year may reduce unpredictability and barriers.
For a young woman who struggles to get to an appointment at a clinic in a town or who can’t keep pills without facing stigma or violence, an injection just twice a year is the option that could keep her free of HIV.
The Gates Foundation and Indian drugmaker Hetero Labs Ltd. are among the groups moving to produce the medication, lenacapavir, which Gilead Sciences Inc. sells in the US for a list price of more than $28,000 annually under the brand name Yeztugo.
Another Indian manufacturer, Dr. Reddy’s Laboratories Ltd., is also stepping in, partnering with Unitaid, the Clinton Health Access Initiative, and Wits RHI to expand supply. Together, these moves will likely accelerate global momentum to make the injection available to millions of people most at risk of HIV.
Team Maverick
Raksha Mantri chairs Parliamentary Consultative Committee meeting for MoD on Border Roads Organisation
BRO is fostering an ecosystem along the borders encompassing national security, developmen…